Abstract
Introduction Hydroxyurea (HU) is the most widely adopted first-line cytoreductive agent for polycythemia vera (PV) patients. Approximately a quarter of PV patients receiving HU develop resistance/intolerance, which has been associated with an increased risk of thrombosis, leukemic transformation, and death. Predictors for HU treatment duration, which may be limited due to resistance/intolerance, remain largely unknown. We conducted an exploratory investigation to identify predictive clinical variables for HU treatment duration in PV patients.
Methods We used a previously described multi-institutional PV database consisting of 527 patients from 10 institutions in the United States. PV patients were retrospectively identified as 1) having been treated with HU, and 2) having at least one follow-up visit after HU initiation. This group was further divided based on HU status at the cutoff time of the last follow-up (continued vs. discontinued). We first looked for variables associated with HU status, using Fisher's exact test for categorical variables and the Kolmogorov-Smirnov test for continuous variables. Next, we conducted a survival analysis to calculate predicted HU treatment duration and to look for variables associated with it, using the log-rank test for categorical variables, Cox proportional-hazard model for continuous variables (HU status was used as the event/censor variable).
Results Of 377 qualified PV patients, 164 were in the HU discontinuation group, and 213 were in the treatment continuation group. Baseline characteristics of these patients are summarized in Table 1.
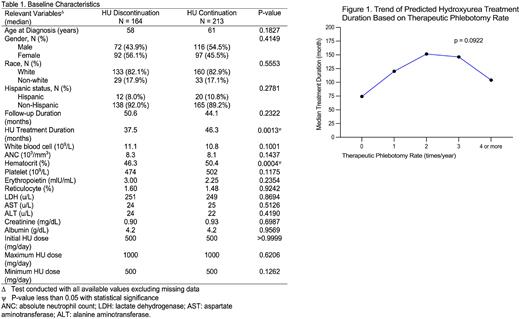
Patients treated with both therapeutic phlebotomy (TP) and HU had a significantly longer estimated duration of HU treatment with a predicted median treatment duration of 107.5 months (n = 284) compared to 74.3 months in those not treated with concurrent TP (n = 93) (HR 1.58 with 95% CI 1.08 to 2.32, p = 0.0190). Further exploration was conducted to identify the association between TP frequency and HU treatment duration. Although lacking statistical significance (p=0.0922), patients who required TP twice a year were found to have the longest predicted HU treatment duration (151.6 months) compared to patients without receipt of TP (74.2 months) or those who required TP once a year (120.1 months), three times a year (146.2 months), and four or more times a year (104.0 months). The trend of predicted HU treatment duration is presented in Figure 1.
Patients with any abnormal karyotype (n = 30) were found to have discontinued HU sooner, with a predicted median treatment duration of 59.8 months compared to 75.4 months in patients with a normal karyotype (n = 102). HR 1.81 with 95% CI 1.01 to 3.25, p = 0.0466.
Baseline hematocrit is the only statistically significant variable in hematologic laboratory results. Although median baseline hematocrit is lower in the HU discontinuation group, it is not associated with longer predicted HU treatment duration in the Cox proportional-hazard regression model. HR 0.99 with 95% CI 0.97 to 1.01, p = 0.2218.
Conclusions In this analysis, we found that PV patients requiring both TP and HU, likely those with prominent myeloproliferation and bone marrow reserve, may remain on HU treatment longer. Normal karyotype was found to be associated with significantly longer predicted HU treatment duration. Baseline hematocrit was lower in HU discontinuation group but not associated with longer predicted HU treatment duration. Prospective studies are necessary to clarify predictors of HU treatment duration and reasons for HU discontinuation.
Disclosures
Podoltsev:CTI BioPharma: Honoraria; PharmaEssentia: Honoraria; Bristol-Myers Squibb: Honoraria; Celgene: Honoraria; Novartis: Honoraria; Pfizer: Honoraria; Cogent Biosciences: Other: Independent Data Review Committee ; AbbVie: Honoraria; Blueprint Medicines: Honoraria; Agios Pharmaceuticals: Honoraria; Incyte: Honoraria; Constellation Pharmaceuticals: Honoraria. Gotlib:Blueprint Medicines Corporation: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Research Funding; PharmaEssentia: Honoraria, Membership on an entity's Board of Directors or advisory committees; Deciphera: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Incyte: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Kartos: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Cogent Biosciences: Consultancy, Research Funding; Allakos: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Heaney:CTI BioPharma: Consultancy, Research Funding; BMS: Research Funding; Blueprint Medicine: Consultancy, Research Funding; Cogent Biosciences: Consultancy, Research Funding; Hoth Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Incyte: Research Funding; Kartos: Research Funding; Novartis: Consultancy, Research Funding; PharmaEssentia: Consultancy; Sierra Oncology: Consultancy, Research Funding. Kuykendall:Morphosys: Other: Research Support; Protagonist: Other: Research Support; BMS: Consultancy, Honoraria, Other: Research Support, Speakers Bureau; Prelude Pharmaceuticals: Other: Research Support; GSK - Sierra Oncology: Consultancy, Honoraria, Other: Research Support, Speakers Bureau; Abbvie: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; Blueprint: Consultancy, Honoraria, Speakers Bureau; Incyte: Consultancy, Honoraria, Speakers Bureau; Imago Biosciences: Consultancy, Honoraria, Speakers Bureau; CTI Biopharma: Consultancy, Honoraria, Speakers Bureau; Pharmaessentia: Consultancy, Honoraria, Speakers Bureau. Fleischman:BMS: Speakers Bureau; CTI: Speakers Bureau; Pharmaessentia: Speakers Bureau; Incyte: Consultancy, Honoraria. Mesa:CTI: Research Funding; Promedior: Research Funding; Incyte: Consultancy, Research Funding; Samus: Consultancy, Research Funding; AbbVie: Research Funding; Genotech: Research Funding; Blueprint: Consultancy; Geron: Consultancy; Roche: Consultancy; Bristol Myers Squibb: Consultancy; Celgene: Research Funding; AOP: Consultancy; Constellation Pharmaceuticals, Inc., a MorphoSys Company: Consultancy, Research Funding; Novartis: Consultancy; LaJolla Pharmaceutical: Consultancy; Sierra Oncology: Consultancy, Research Funding; Gilead: Research Funding; Imago: Research Funding. Yacoub:Gilead: Consultancy; AbbVie: Consultancy; Apellis: Consultancy; Agios: Consultancy; Acceleron Pharma: Consultancy; Notable Labs: Consultancy; Stemline Therapeutics: Research Funding; Servier: Consultancy; Novartis: Consultancy; Pfizer: Consultancy; PharmaEssentia: Consultancy; CTI Pharma: Consultancy; Incyte: Consultancy, Speakers Bureau. Mascarenhas:Prelude Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene/BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; CTI BioPharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Merus: Research Funding; PharmaEssentia: Consultancy, Research Funding; Geron: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; GSK: Consultancy; Kartos: Consultancy, Research Funding; Sierra Oncology: Consultancy; Constellation Pharmaceuticals, Inc., a MorphoSys Company: Consultancy; AbbVie: Consultancy, Research Funding; Galecto: Consultancy; Forbius: Research Funding; Merck: Research Funding; Janseen: Research Funding; Karyopharm: Consultancy, Membership on an entity's Board of Directors or advisory committees; Imago: Consultancy; Roche: Consultancy, Research Funding; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Shammo:BMS: Consultancy, Honoraria, Research Funding, Speakers Bureau; Apellis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau; Astra Zeneca: Research Funding; Takeda: Consultancy, Current equity holder in private company, Current equity holder in publicly-traded company, Honoraria, Research Funding; Abbvie: Current equity holder in private company, Research Funding; Baxter: Current holder of stock options in a privately-held company; Sanofi: Consultancy, Honoraria, Speakers Bureau; NS Pharma: Membership on an entity's Board of Directors or advisory committees; Alexion: Consultancy, Honoraria, Research Funding, Speakers Bureau; Kartos Pharma: Research Funding; Incyte: Consultancy, Honoraria, Research Funding, Speakers Bureau; CTI Pharma: Research Funding; Stemline Therapeutics: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal